|
4/15/2024 0 Comments Periodic table molar mass of fe(The above picture of the periodic system is interactive - no need to download, just click on an element. Now available: history of the periodic tableĬhoose elements by name, by atomic number, by symbol, by massĬlick here for the history of the periodic table.Ĭlick here to download a PDF version from that periodic table An interactive, printable extended version of the Periodic table of chemical elements of Mendeleev (who invented the periodic table). Phone: +31 152 610 900 chemical element contains a link to a page that explains its chemical properties, health effects, environmental effects, application data, an image and also information of the history/inventor of each element.Separation and Concentration Purification Request.Plant Inspection & Process Optimalisation.Select the correct mass for each of the samples described below. Feel free to contact me if you have any questions. Use the periodic table to determine how many atoms of phosphorus (P) are in a sample that has a mass of 172.90 g. Molar Masses were found using a periodic table: Molar Mass of Fe 55.84 g/mol. However, even these small quantities are required for the body to function properly. Since the question gives the grams of Fe with 3 sig figs, our answer should also be given in 3 sig figs. These last three metals are present in the body in very small quantities. Since protons and neutrons contribute the. In terms of weight, one unified atomic mass unit is equivalent to one-twelfth of the mass of a Carbon-12 atom at rest. Unified atomic mass units are commonly used to quantify atomic mass. Cobalt is a necessary component of vitamin B-12, a vital nutrient. The atomic mass of an element is a measure of the average mass of its atoms, expressed in Atomic Mass Units (AMU). Manganese is needed for the body to metabolize oxygen properly. Copper is also needed for several proteins to function properly in the body. Zinc is needed for the body’s immune system to function properly, as well as for protein synthesis and tissue and cell growth. Other transition metals have important functions in the body, despite being present in low amounts. It is the presence of this particular transition metal in your red blood cells that allows you to use the oxygen you inhale. Each hemoglobin molecule has four iron atoms, which act as binding sites for oxygen. Simply type in the remaining values and watch it do all the work for you. You can also use this molarity calculator to find the mass concentration or molar mass. The crucial atom in the hemoglobin protein is iron. Substitute the known values to calculate the molarity: molarity 5 / (1.2 × 36.46) 0.114 mol/l 0.114 M. Hemoglobin is a relatively large molecule, with a mass of about 65,000 u. Hemoglobin combines with oxygen and carbon dioxide, transporting these gases from one location to another in the body. The critical part of the red blood cell is a protein called hemoglobin. Most periodic tables provide additional data (such as atomic mass) in a box that contains each element’s symbol. Without red blood cells, animal respiration as we know it would not exist. The periodic table is one of the cornerstones of chemistry because it organizes all the known elements on the basis of their chemical properties. Red blood cells are cells that transport oxygen from the lungs to cells of the body and then transport carbon dioxide from the cells to the lungs. Add them together: add the results from step 3 to get the total molar mass of the compound. Calculate molar mass of each element: multiply the atomic mass of each element by the number of atoms of that element in the compound. 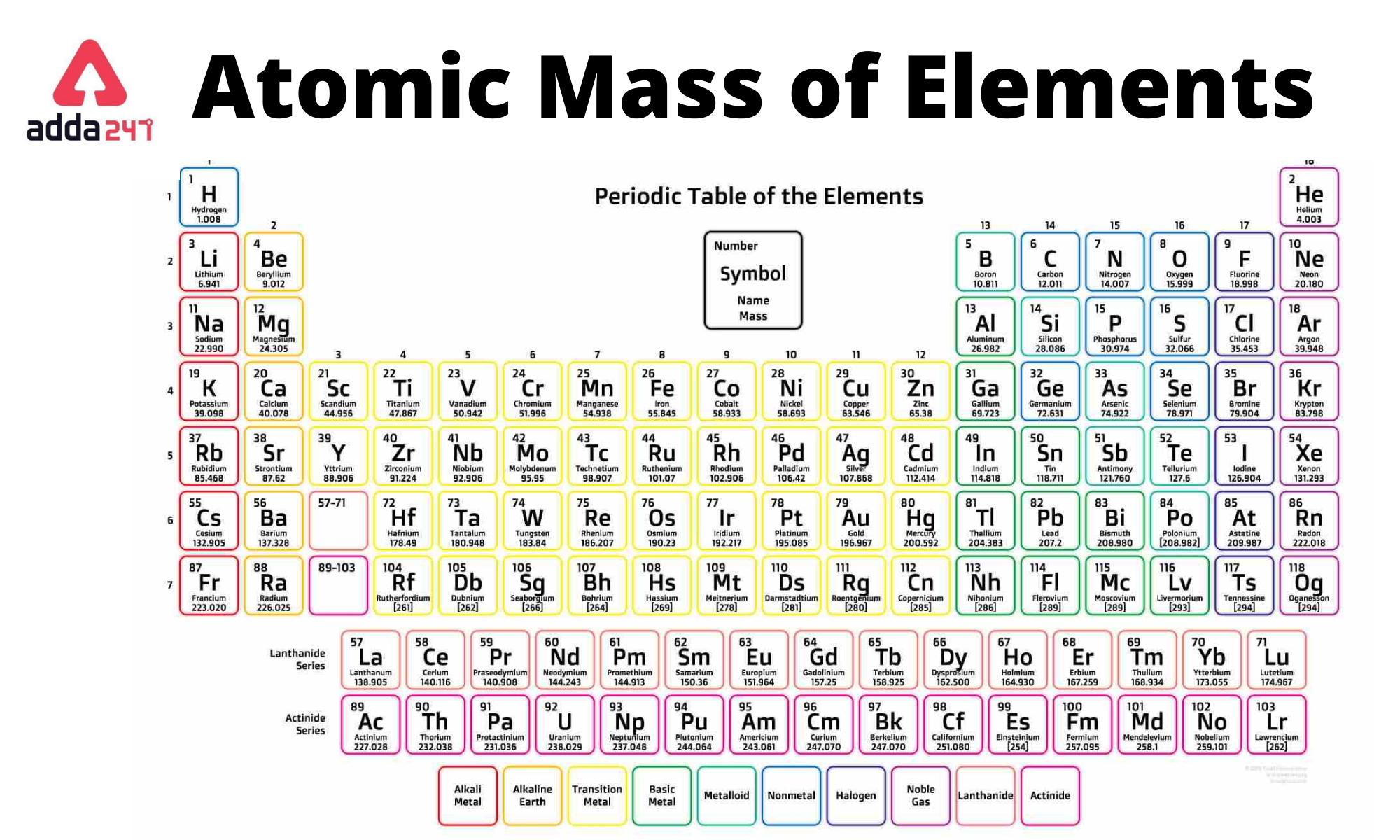
Iron is a transition metal and the chemistry of iron makes it a key component in the proper functioning of red blood cells. The atomic mass is usually found on the periodic table and is given in atomic mass units (amu). Because iron has relatively massive atoms, it would appear even lower on a list organized in terms of percent by atoms rather than percent by mass. The most abundant non-main group element is iron, at 0.006 percentage by mass. :max_bytes(150000):strip_icc()/PeriodicTable-58b5d9253df78cdcd8d03ebb.jpg) 
Most of the elemental composition of the human body consists of main group elements. To Your Health: Transition Metals in the Body Each group is located in a different part of the periodic table. Elements are either metals, nonmetals, or semimetals.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
